 We propose that impaired function of the PV inhibitory circuit contributes to epileptic seizures in knock-in mice. Finally, we show that Na v1.1 is involved in sustained high-frequency firing of neocortical fast-spiking interneurons. In the hippocampus, Na v1.1 is also predominantly distributed within somata and axons of PV interneurons, whereas pyramidal neurons express Na v1.1 at extremely low levels. Na v1.1 is predominantly found at the axon initial segments of parvalbumin-positive (PV) interneurons. Moreover, we describe a novel form of Na v1.1 localization in the developing neocortex. We show the absence of truncated mutant Na v1.1 in their brains, and the resultant Na v1.1 haploinsufficiency causes epileptic seizures in these mice. We report here the generation and characterization of knock-in mice carrying a truncation mutation in the Scn1a gene identical to the human SMEI mutation. However, a reexamination of Na v1.1 distribution and localization in the brain is required to resolve why the loss of Na v1.1 affects only inhibitory, but not pyramidal, neurons when both cell types express the channel.  
(2006) generated Na v1.1-null mice exhibiting spontaneous seizures and whole-cell sodium currents that were significantly reduced in isolated GABAergic interneurons but not in pyramidal cells from hippocampus, suggesting that loss of Na v1.1 might specifically decrease inhibition resulting in epilepsy. Because SMEI mutations are mainly heterozygous nonsense or frame-shift mutations leading to predicted loss of Na v1.1 function, Na v1.1 haploinsufficiency has been implicated in SMEI pathology ( Sugawara et al., 2003 Meisler and Kearney, 2005 Mulley et al., 2005 Yamakawa, 2005). The first seizure is typically a unilateral or generalized tonic-clonic or clonic seizure often, but not always, associated with fever and is subsequently followed by additional generalized and partial seizures, ataxia, and mental decline. SMEI begins in children under 1 year of age, who otherwise develop normally before disease onset. SMEI, or Dravet syndrome, is the most severe and intractable form of the SCN1A-associated epileptic disorders (Online Mendelian Inheritance in Man number 607208). To date, >100 heterozygous mutations of human SCN1A gene encoding Na v1.1 have been reported in several human epileptic disorders, namely, generalized epilepsy with febrile seizure plus (GEFS+), intractable childhood epilepsy with generalized tonic-clonic seizures (ICEGTC), and severe myoclonic epilepsy in infancy (SMEI) ( Escayg et al., 2000 Claes et al., 2001 Fujiwara et al., 2003). The α type I sodium channel (Na v1.1) has been reported to be expressed in the brain, in which it is localized to somata and dendrites of neurons ( Westenbroek et al., 1989 Gong et al., 1999). Four α and four β subunits have been identified in the mammalian brain. The pore-forming α subunits also serve as voltage sensors, whereas the accessory β subunits modulate the voltage dependence and kinetics and cellular localization of the α subunits. 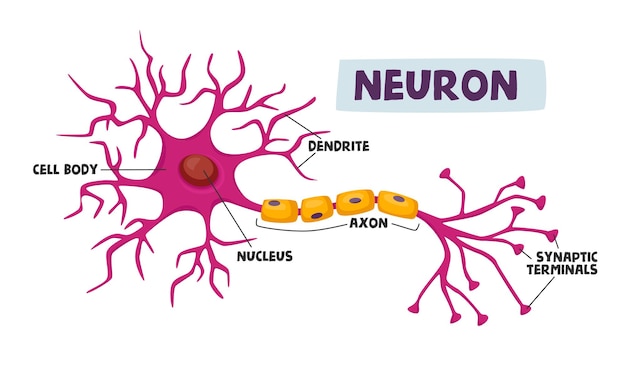
These channels are heteromultimeric protein complexes consisting of one α and one or two β subunits ( Catterall, 2000). Voltage-gated sodium channels are essential for the generation and propagation of action potentials in electrically excitable tissues, such as brain, muscle, and heart. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
